
Learn more about hemophilia A
For more information about hemophilia, visit these Web pages, or read these publications: the National Hemophilia Foundation website (https://www.hemophilia.org/About-Us/Fast-Facts, https://www.hemophilia.org/Bleeding-Disorders/Types-of-Bleeding-Disorders/Hemophilia-A, https://www.hemophilia.org/Bleeding-Disorders/Types-of-Bleeding-Disorders/Hemophilia-B), and Triplett DA. Coagulation and bleeding disorders: review and update. Clin Chem. 2000;46(8 Part 2):1260-1269.
It is estimated that more than 400,000 people in the world have hemophilia
Hemophilia A occurs in 1 in 5000 male births and is about 4 times as common as hemophilia B. The number of people with hemophilia in the United States is estimated to be about 20,000. Approximately 75% of people with hemophilia around the world receive inadequate treatment or have no access to treatment.
What is hemophilia?
Hemophilia is a bleeding disorder that affects the way blood clots. People born with hemophilia are missing some or all of a type of protein called clotting factor. The 2 main types of hemophilia are hemophilia A and hemophilia B. People with hemophilia A are missing some or all of the clotting factor VIII. People with hemophilia B are missing some or all of a different clotting factor called factor IX.
Clotting factors are important because they help in the clotting process. When a blood vessel is cut or injured, clotting factors help platelets (which are cellular fragments made from your bone marrow) form a plug in the wound that stops the bleeding.
Why do mostly males have hemophilia A?
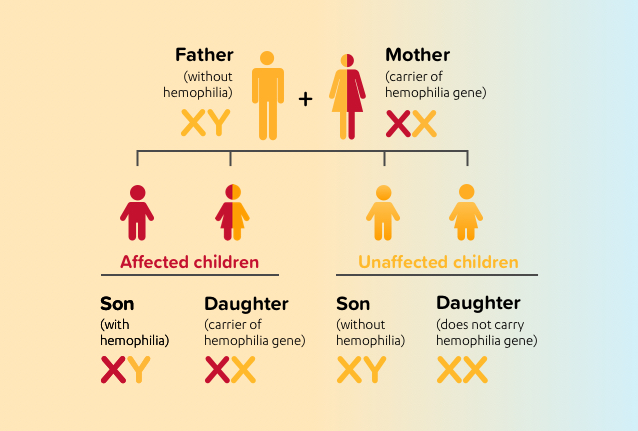
Hemophilia A is caused by a defective gene located on the X chromosome. Females have 2 X chromosomes, and males have 1 X chromosome and 1 Y chromosome. The defective gene is passed down from a mother (who carries, but is unaffected by, the gene) to her son. Females are unaffected because they have 2 copies of the X chromosome, so if the factor VIII gene on an X chromosome is defective, the gene on the other X chromosome can make enough factor VIII. Because males have only 1 X chromosome and 1 Y chromosome, if their only copy of factor VIII gene on the X chromosome is defective, they will have hemophilia A.
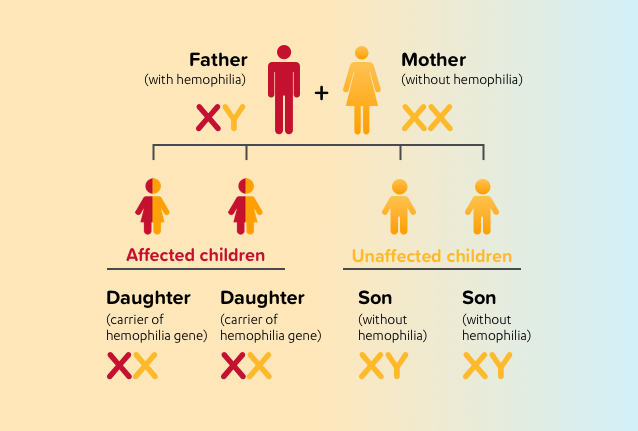
Because females get 1 X chromosome from their mother, and 1 X chromosome from their father, a father who has hemophilia will pass the defective X chromosome to his daughters, and his daughters will be carriers. Because males get their X chromosome from their mother, they will not inherit the defective X chromosome, and therefore males born to a father with hemophilia and a mother who is not a carrier will not be affected.
Important Safety Information
ALPHANATE is contraindicated in patients who have manifested life-threatening immediate hypersensitivity reactions, including anaphylaxis, to the product or its components.
Anaphylaxis and severe hypersensitivity reactions are possible with ALPHANATE. Discontinue use of ALPHANATE if hypersensitivity symptoms occur, and initiate appropriate treatment.
Development of procoagulant activity-neutralizing antibodies (inhibitors) has been detected in patients receiving FVIII-containing products. Carefully monitor patients treated with AHF products for the development of FVIII inhibitors by appropriate clinical observations and laboratory tests.
Thromboembolic events have been reported with AHF/VWF complex (human) in VWD patients, especially in the setting of known risk factors.
Intravascular hemolysis may occur with infusion of large doses of AHF/VWF complex (human).
Rapid administration of a FVIII concentrate may result in vasomotor reactions.
Because ALPHANATE is made from human plasma, it may carry a risk of transmitting infectious agents, eg, viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent, despite steps designed to reduce this risk.
Monitor for development of FVIII and VWF inhibitors. Perform appropriate assays to determine if FVIII and/or VWF inhibitor(s) are present if bleeding is not controlled with expected dose of ALPHANATE.
The most frequent adverse drug reactions reported with ALPHANATE in >1% of infusions were pruritus, headache, back pain, paresthesia, respiratory distress, facial edema, pain, rash, and chills.
Please see full Prescribing Information for ALPHANATE.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1.800.FDA.1088.


